Resume Skills and Keywords for Quality Assurance Microbiologist
A Quality Assurance Microbiologist plays a vital role in ensuring the safety, quality, and compliance of pharmaceutical, food, and cosmetic products through rigorous microbiological testing and analysis. They monitor production environments, validate sterilisation processes, and conduct microbial limit tests, endotoxin tests, and environmental monitoring. By applying their expertise in microbiology, aseptic techniques, and quality systems such as GMP, GLP, and ISO standards, they help prevent contamination and maintain product integrity. Working closely with production, R&D, and regulatory teams, Quality Assurance Microbiologists ensure that all products meet strict microbiological and quality specifications before release—safeguarding both consumer health and brand reputation.
Skills required for a Quality Assurance-Microbiologist role:
- Total Viable Count (TVC)
- Yeast & Mold Count
- Microbial Limit
- Environmental Monitoring
- Media Preparation & Culture Handling
- Equipment Handling
- Validation & Sterilization
- Good Laboratory Practices (GLP)
- Regulatory Compliance
- Team Management
What recruiters look for in a Quality Assurance-Microbiologist's resume:
- Bachelor’s or Master’s degree in Microbiology, Biotechnology, Biochemistry, or a related life science field.
- Strong foundation in microbial physiology, aseptic techniques, and contamination control.
- Hands-on experience in microbiological testing such as sterility testing, microbial limit testing, endotoxin (LAL) testing, and environmental monitoring.
- Proficiency in using laboratory instruments like autoclaves, incubators, laminar airflow cabinets, and colony counters.
- Knowledge of GMP, GLP, ISO 9001/13485 standards, and regulatory compliance related to microbiological quality assurance.
- Experience in media preparation, validation, and equipment qualification for microbiology laboratories.
- Familiarity with documentation practices, including SOP preparation, deviation reporting, and CAPA management.
What can make your Quality Assurance-Microbiologist resume stand out:
A strong summary that demonstrates your skills, experience and background in quality assurance
- A passionate QA Microbiologist with a strong foundation in microbiological testing, quality control, and laboratory practices. Skilled in aseptic techniques, media preparation, and microbial analysis of raw materials and finished products. Knowledgeable in GMP, GLP, and ISO standards with hands-on exposure to environmental monitoring and documentation.
Targeted job description
- Lead the design, implementation, and continuous improvement of microbiological quality assurance processes to ensure compliance with regulatory and organisational standards.
- Conduct routine microbiological testing including sterility, microbial limit, and endotoxin (LAL) tests to ensure product and environmental quality.
- Oversee environmental monitoring programs, including air, surface, and water testing, and ensure timely corrective actions for deviations or contamination events.
- Utilise validated microbiological methods and instruments to perform identification, enumeration, and characterisation of microorganisms in raw materials, in-process, and finished products.
- Collaborate with cross-functional teams in production, R&D, and regulatory affairs to support process validation, equipment qualification, and product release decisions.
Related academic background
- M.Sc Microbiology at Dr. B. Lal. Institute of Biotechnology, Jaipur | 2023
- B.Sc Biotechnology at Dr. B. Lal. Institute of Biotechnology, Jaipur | 2021
- ISO 9001:2015 – Quality Management Systems (QMS)
- ISO 17025 – Testing & Calibration Laboratories
- Internal Auditor / Lead Auditor Training (ISO 9001 or ISO 17025)
Sample Resume of Quality Assurance Microbiologist in Text Format
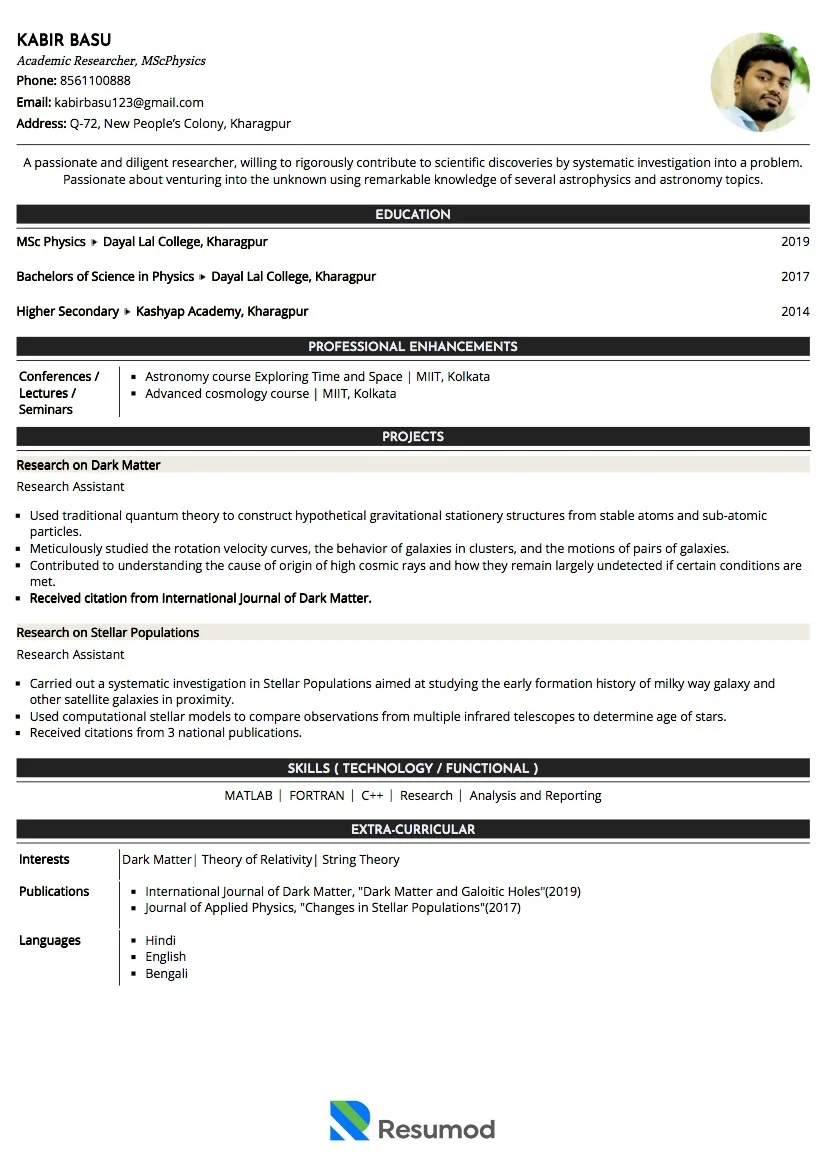
ANMOL VERMA
Quality Assurance-Microbiologist
+91-XXXXXXXXXX | support@resumod.co |Pune, India
SUMMARY
A passionate QA Microbiologist with a strong foundation in microbiological testing, quality control, and laboratory practices. Skilled in aseptic techniques, media preparation, and microbial analysis of raw materials and finished products. Knowledgeable in GMP, GLP, and ISO standards with hands-on exposure to environmental monitoring and documentation.
EMPLOYMENT HISTORY
Quality Assurance-Microbiologist at Sun Pharma Ltd. from May 2024 - Present, Pune
- Performing microbiological testing of raw materials, in-process samples, and finished products (e.g., total microbial count, yeast and mold count, pathogen detection).
- Conducting sterility tests, endotoxin tests, and bioburden assessments as per regulatory guidelines.
- Evaluating microbial limits and ensuring results meet established specifications.
- Identifying and reporting potential contamination sources and deviations.
- Recommending corrective and preventive actions (CAPA) for microbiological deviations.
Trainee Microbiologist at Dr. Lal Path Labs from Aug 2023 - Apr 2024, Pune
- Assisted in performing routine microbiological testing of raw materials, in-process samples, and finished products.
- Helped in preparing culture media, reagents, and sterilization of laboratory equipment.
- Followed Standard Operating Procedures (SOPs) and ensured compliance with GMP/GLP standards.
- Adhered to company safety policies and laboratory hygiene practices.
- Reported any deviations, contamination, or abnormalities to the supervisor immediately.
CERTIFICATIONS
- ISO 9001:2015 – Quality Management Systems (QMS)
- ISO 17025 – Testing & Calibration Laboratories
- Internal Auditor / Lead Auditor Training (ISO 9001 or ISO 17025)
EDUCATION
- M.Sc Microbiology at Dr. B. Lal. Institute of Biotechnology, Jaipur | 2023
- B.Sc Biotechnology at Dr. B. Lal. Institute of Biotechnology, Jaipur | 2021
SKILLS
Total Viable Count (TVC) | Yeast & Mold Count | Microbial Limit | Environmental Monitoring | Media Preparation & Culture Handling | Equipment Handling | Validation & Sterilization | Good Laboratory Practices (GLP) | Regulatory Compliance | Team Management
LANGUAGES
English
Hindi


 10031
10031